New Delivery for Huperzine A El Salvador
New Delivery for Huperzine A El Salvador Detail:
[Latin Name]Huperzia serratum
[Source] Huperziceae whole herb from China
[Appearance]Brown to white
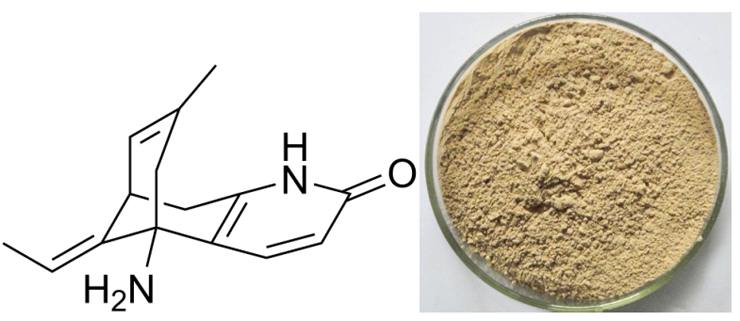
[Ingredient]Huperzine A
[Specification]Huperzine A 1% – 5%, HPLC
[Solubility] Soluble in chloroform, methanol, ethanol, slightly soluble in water
[Particle size] 80 Mesh
[Loss on drying] ≤5.0%
[Heavy Metal] ≤10PPM
[Pesticide residue] EC396-2005, USP 34, EP 8.0, FDA
[Storage] Store in cool & dry area, keep away from the direct light and heat.
[Shelf life] 24 Months
[Package] Packed in paper-drums and two plastic-bags inside.
[What is Huperzine A]
Huperzia is a type of moss that grows in China. It is related to club mosses (the Lycopodiaceae family) and is known to some botanists as Lycopodium serratum . The whole prepared moss was used traditionally. Modern herbal preparations use only the isolated alkaloid known as huperzine A. Huperzine A is an alkaloid found in huperzia that has been reported to prevent the breakdown of acetylcholine, an important substance needed by the nervous system to transmit information from cell to cell. Animal research has suggested that huperzine A’s ability to preserve acetylcholine may be greater than that of some prescription drugs. Loss of acetylcholine function is a primary feature of several disorders of brain function, including Alzheimer’s disease . Huperzine A may also have a protective effect on brain tissue, further increasing its theoretical potential for helping reduce symptoms of some brain disorders.
[Function] Used in alternative medicine, huperzine A has been found to act as a cholinesterase inhibitor, a type of medicine used to prevent the breakdown of acetylcholine (a chemical essential to learning and memory).
Not only used as a treatment for Alzheimer’s disease, huperzine A is also said to enhance learning and memory and to protect against age-related cognitive decline.
In addition, huperzine A is sometimes used to boost energy, increase alertness, and aid in the treatment of myasthenia gravis (an autoimmune disorder that affects the muscles).
Product detail pictures:

Related Product Guide:
The pretty loaded projects management experiences and one to a person support model make the high importance of business enterprise communication and our easy understanding of your expectations for New Delivery for Huperzine A El Salvador , The product will supply to all over the world, such as: Peru, Sri Lanka, Bulgaria, We have been in operation for more than 10 years. We are dedicated to quality products and consumer support. We currently own 27 product utility and design patents. We invite you to visit our company for a personalized tour and advanced business guidance.
Expand your vocabulary and learn how to say new words:
https://www.dictionaryvoice.com/How_To_Pronounce_Polysaccharides.html
Please leave a Like, a Comment, and Share.
Bookmark us and share:
https://www.dictionaryvoice.com
Twitter:
https://twitter.com/DictionaryVoice
Facebook:
https://www.facebook.com/pages/Dictionary-Voice/750369141710497
More Pronunciations:
1) How to Pronounce Polysaccharides
https://www.dictionaryvoice.com/How_To_Pronounce_Polysaccharides.html
QUFU, China, Jan. 7, 2013 /PRNewswire/ — Sunwin Stevia International, Inc. “Sunwin Stevia” (SUWN) one of the top global providers of high quality stevia extracts including Rebaudioside A 98, announced today that its facilities are now capable of producing A3-99 stevia products. The addition of A3-99 to its commercial production capabilities is part of its previously announced stevioside expansion project.
A3-99 extracts are among the highest quality stevioside extracts produced in the world and are used in the pharmaceutical and food and beverage industries. Sunwin Stevia has completed testing for this production process making it one of a select few producers of A3-99 in China. Management expects to begin delivering shipments of the A3-99 to Zhejiang Huisong Pharmaceuticals, a pharmaceutical customer in China, in the fourth quarter of fiscal 2013 which begins in February 2013. Sunwin also intends to seek patents in China for its A3-99 production methodology.
“We are excited to add A3-99 to our commercial product offerings making Sunwin Stevia one of a select few suppliers capable of producing this extract on a large scale. Our recent facility upgrade has substantially increased our overall capabilities and we anticipate further additions to our production capabilities as we continue testing and trial production runs,” commented Dongdong Lin, CEO of Sunwin Stevia. “We believe our technology is state of the art and we intend to seek a patent for our methodologies. We look forward to actively marketing our A3-99 stevia in China and eventually in the international markets in the coming quarters.”
About Sunwin Stevia International, Inc.
Sunwin Stevia International, Inc. engages in the areas of zero calorie, all natural sweeteners (Sunwin Stevia™ Extracts). As an industry leader in agricultural processing, Sunwin has built an integrated global firm with the sourcing and production capabilities to meet the needs of consumers throughout the world. For more info about Sunwin, please visit https://www.sunwininternational.com
Safe Harbor Statement
Sunwin Stevia International, Inc. is hereby providing cautionary statements identifying important factors that could cause our actual results to differ materially from those projected in forward-looking statements (as defined in such act). Any statements that are not historical facts and that express, or involve discussions as to, expectations, beliefs, plans, objectives, assumptions or future events or performance (often, but not always, indicated through the use of words or phrases such as “will likely result,” “are expected to,” “will continue,” “is anticipated,” “estimated,” “intends,” “plans,” “believes” and “projects”) may be forward-looking and may involve estimates and uncertainties which could cause actual results to differ materially from those expressed in the forward-looking statements. These statements include, but are not limited to, our ability to obtain patents, future production capabilities, our technology and the market for our products, and other risk factors impacting our company, some of which may be beyond our control. We caution that the factors described herein could cause actual results to differ materially from those expressed in any forward-looking statements we make and that investors should not place undue reliance on any such forward-looking statements. Further, any forward-looking statement speaks only as of the date on which such statement is made, and we undertake no obligation to update any forward-looking statement to reflect events or circumstances after the date on which such statement is made or to reflect the occurrence of anticipated or unanticipated events or circumstances. New factors emerge from time to time, and it is not possible for us to predict all of such factors. Further, we cannot assess the impact of each such factor on our results of operations or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward-looking statements. This press release is qualified in its entirety by the cautionary statements and risk factor disclosure contained in our Securities and Exchange Commission filings, including our Annual Report on Form 10-K for the fiscal year ended April 30, 2012.
Contact:
Dore Perler
U.S. Representative
954-232-5363
ir@sunwininternational.com
This supplier's raw material quality is stable and reliable, has always been in accordance with the requirements of our company to provide the goods that quality meet our requirements.