Popular Design for Flaxseed Extract Factory for Canberra
Popular Design for Flaxseed Extract Factory for Canberra Detail:
[Latin Name] Linum Usitatissimum L.
[Plant Source] from China
[Specifications]SDG20% 40% 60%
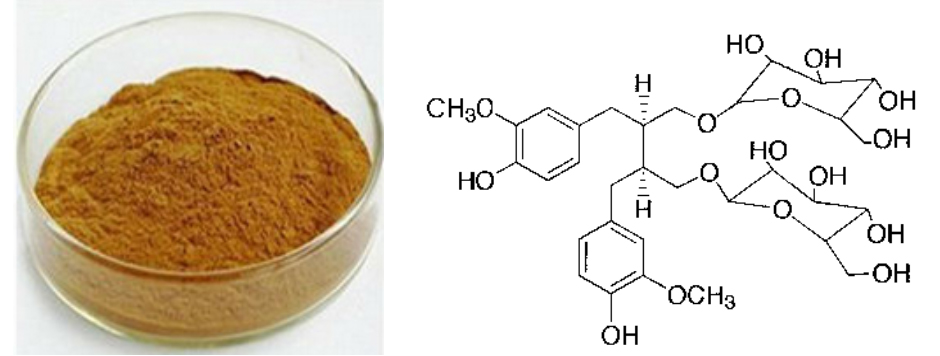
[Appearance] yellow brown powder
Plant Part Used:Seed
[Particle size] 80 Mesh
[Loss on drying] ≤5.0%
[Heavy Metal] ≤10PPM
[Storage] Store in cool & dry area, keep away from the direct light and heat.
[Shelf life] 24 Months
[Package] Packed in paper-drums and two plastic-bags inside.
[Net weight] 25kgs/drum
Product description:
Flaxseed extract is a kind of plant ligan most notably found in flaxseed. Secoisolariciresinol diglycoside, or SDG is existed as its main bioactive components. SDG is classified as a phytoestrogen since it is a plant-derived, nonsteroid compound that possesses estrogen-like activity. Flaxseed extract SDG has weak estrogenic activity, when intake as food it will be trasfer to flax ligan which have same structure with estrogens.The level of SDG in flaxseed typically varies between 0.6% and 1.8%. Flaxseed extract powder SDG can reduce the blood lipid, cholesterin and triglyceride, it can also prevent for apoplexy, hyperension, blood clots, arteriosclerosis and arrhythmia. In addition, flax seed extract powder SDG is benificial for diabetes and CHD.
Main Function:
1.Flaxseed extract used to lose weight. Can burn surplus fat of Body;
2.Flaxseed extract will reduce allergic reaction, reduce asthma, improve arthritis;
3.Flaxseed extract with the function of improving female menstrual period syndrome;
4.Flaxseed extract can reduce the bad influence of hazardous chemicals produced when under pressure, control Stress, reduce depression and insomnia;
5.Flaxseed extract will improve skin fat content, moisten the skin smooth, soft and flexible, make the skin breath and sweat to normal, to mitigate various skin problems.
Product detail pictures:

Related Product Guide:
carry on to boost, to guarantee products excellent in line with market and consumer standard specifications. Our enterprise has a quality assurance system are actually established for Popular Design for Flaxseed Extract Factory for Canberra , The product will supply to all over the world, such as: Swedish, South Korea, Kuwait, we always keep our credit and mutual benefit to our client, insist our high quality service to moving our clients. always welcome the our friends and clients to come and visit our company and guid our business, if you are interested in our products, you can also submit your purchase information online, and we will contact you immediately, we keep our highly sincere cooperation and wish everything in your side are all well.
About Sunwin Stevia International, Inc.
Sunwin Stevia International, Inc. engages in the areas of zero calorie, all natural sweeteners (Sunwin Stevia™ Extracts). As an industry leader in agricultural processing, Sunwin has built an integrated global firm with the sourcing and production capabilities to meet the needs of consumers throughout the world. For more info about Sunwin, please visit https://www.sunwininternational.com
Safe Harbor Statement
Sunwin Stevia International, Inc. is hereby providing cautionary statements identifying important factors that could cause our actual results to differ materially from those projected in forward-looking statements (as defined in such act). Any statements that are not historical facts and that express, or involve discussions as to, expectations, beliefs, plans, objectives, assumptions or future events or performance (often, but not always, indicated through the use of words or phrases such as “will likely result,” “are expected to,” “will continue,” “is anticipated,” “estimated,” “intends,” “plans,” “believes” and “projects”) may be forward-looking and may involve estimates and uncertainties which could cause actual results to differ materially from those expressed in the forward-looking statements. These statements include, but are not limited to, our expectations regarding the costs of development of stevia based products and our belief regarding the future growth of our stevia distribution business and our expectations regarding future sales and distribution channel development. We caution that the factors described herein could cause actual results to differ materially from those expressed in any forward-looking statements we make and that investors should not place undue reliance on any such forward-looking statements. Further, any forward-looking statement speaks only as of the date on which such statement is made, and we undertake no obligation to update any forward-looking statement to reflect events or circumstances after the date on which such statement is made or to reflect the occurrence of anticipated or unanticipated events or circumstances. New factors emerge from time to time, and it is not possible for us to predict all of such factors. Further, we cannot assess the impact of each such factor on our results of operations or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward-looking statements. This press release is qualified in its entirety by the cautionary statements and risk factor disclosure contained in our Securities and Exchange Commission filings, including our Annual Report on Form 10-K for the fiscal year ended April 30, 2012.
Contact:
Dore Perler
U.S. Representative
954-232-5363
ir@sunwininternational.com
Customer service staff and sales man are very patience and they all good at English, product's arrival is also very timely, a good supplier.