Trending Products Dandelion root extract Wholesale to Japan
Trending Products Dandelion root extract Wholesale to Japan Detail:
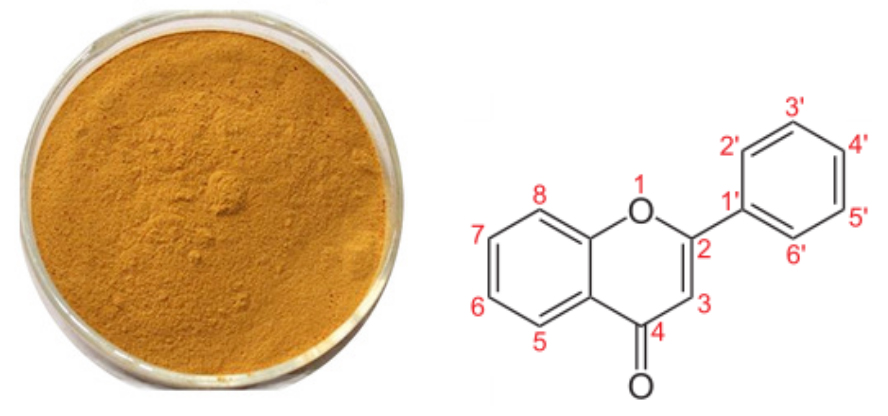
[Latin Name] Taraxacum officinale
[Plant Source] from China
[Specifications] Flavones 3%-20%
[Appearance] Brown fine powder
Plant Part Used:Root
[Particle size] 80 Mesh
[Loss on drying] ≤5.0%
[Heavy Metal] ≤10PPM
[Storage] Store in cool & dry area, keep away from the direct light and heat.
[Shelf life] 24 Months
[Package] Packed in paper-drums and two plastic-bags inside.
[Net weight] 25kgs/drum
[Function]
(1) It is a general stimulant to the system, but especially to the urinary organs, and is chiefly used in kidney and liver disorders;
(2) Dandelion is also used as a remedy for hemorrhoids, gout, rheumatism, eczema, other skin conditions, and diabetes.
(3) Dandelion is used to treat chronic ulcers, stiff joints, and tuberculosis. It is also used to induce milk production in nursing mothers and to soothe inflamed breast tissue.
[Pharmacological effects]
(1) the antibacterial action: made of injection to extract the dandelion staphylococcus aureus and have strong hemolytic streptococcus pneumoniae, to kill, meningococci, diphtheria bacili, pseudomonas aeruginosa, proteus, dysenteric bacili, typhoid bacillus and card he also must kill staphylococcus, fungi, viruses, and some of the leptospira bacterium.
(2)other function. Advantageous bravery,diuresis and bitter soa, mild diarrhea inferior.
[Applications]
Dandelions extract injection, decoction, tablet, syrup, etc for a variety of infection are dampness.the curative effects, including the upper respiratory tract infection and chronic bronchitis, pneumonia, contagious hepatitis, urinary tract infection, surgical disorders, surgery, dermatology inflammation and sepsis inflammation, typhoid, biliary feeling, mumps, etc.
Product detail pictures:

Related Product Guide:
"Sincerity, Innovation, Rigorousness, and Efficiency" is definitely the persistent conception of our corporation to the long-term to establish alongside one another with customers for mutual reciprocity and mutual profit for Trending Products Dandelion root extract Wholesale to Japan , The product will supply to all over the world, such as: Philadelphia, Malaysia, Nairobi, As an experienced factory we also accept customized order and make it same as your picture or sample specifying specification and customer design packing. The main goal of the company is to live a satisfactory memory to all the customers, and establish a long term win-win business relationship. For more information, please contact us. And It is our great pleasure if you like to have a personally meeting in our office.
Know the Natural Male Enhancement methods and procedure to improve sexual health of men – https://www.performanceinsiders.com/male-enhancement-natural.html
Why wait Order now :
https://myrainoffice.com/enroll/self_…
Information :
https://www.myrainlife.com/godsendwell…
https://www.facebook.com/rainsoullife
Rain Soul Powerful Antioxidants & Essential Fatty Acids to energize, revitalize, detoxify your mind, body & soul. Black cumin seed, Black raspberry seed, Grape seed, Ribose-D, Resveratrol.
富 含 抗 氧 化, 人 体 内 必 需 脂 肪 酸, 帮 助 人 脑 和 身 体 排 毒. 古 书 记 载 黑 小 茴 香 除 了 无 法 起 死 回 生, 种 籽 可 做 为 每 种 疾 病 的 治 疗 药 物.
Dr Otto Heinrich Warburg (Germany)
Nobel Prize in Medicine 1931 : Biochemist
The body requires special fats that, among other important functions. make it possible for sufficient oxygen to reach the cells via the cellular membranes. Which are the key, These special fats are highly oxygen-absorbing. Called Essential Fatty Acids, or EFAs, these special fats must be supplied from outside the body every day.
Black raspberry seed :
Contain antioxidant-like phytonutrients including ellagitannins and anthocyanins. These may help fight viruses, inflammation, and a number of other serious health conditions. Antioxidants seek out and eliminate cell-damaging substances, called free radicals. Free radicals can occur naturally in the body or from exposure to environmental toxins. Although ellagitannins exist in most berries, raspberries contain the the most potent levels.
葡萄籽 :
葡萄籽具有类黄酮丰富的来源叫做花青素。这是为脑健康重要,由于其自由基淬抗氧化剂和胶原保护效果。原花色素也已显示出明显的延迟的脂质过氧化的发生,并有效地螯合铁离子。自由铁离子是一些过氧化脂质,过氧化氢和羟自由基产生的最有力的推动者。这是一个主要因素帕金森病,在老年人中最常见的神经疾病之一的发病机制。
Grape seed :
Grape seeds have an abundant source of flavonoids called proanthocyanidins. This is important for brain health due to their free radical-quenching antioxidant and collagen-protecting effects. Proanthocyanidins have also been shown to noticeably delay the onset of lipid peroxidation and to effectively chelate iron ions. Free iron ions are some of the most powerful promoters of lipid peroxide, hydrogen peroxide and hydroxyl radical production. This is a major contributor to the pathogenesis of Parkinsons disease, one of the most common neurological diseases in older individuals.
Ribose-D :
Ribose-D is a five-carbon sugar found in every cell in our bodies that combines with oxygen and ATP (adenosine triphoshate) to give energy to each cell. Ribose is also present in RNA (ribonucleic acid), which is one of the main information carriers of living organisms. Oxidative stress, as measured by free radical damage to cells, leads to systematic inflammation. Fortunately, our bodies handle this problem daily. However, if our bodies experience an abnormal increase in free radicals due to exercise, daily stress, excess smoking, excess saturated fat intake, depressed immune system, aging, etc., the body becomes fatigued and less efficient in producing Ribose-D to replenish cellular energy. Because of this, we need to supplement with Ribose-D in order to regenerate the cells energy system.
Grape Extracts Resveratrol :
Grape skin extract contains trans-resveratrol which is considered a natural antibiotic that can fight bacterial diseases. Resveratrol may help protect the body from various diseases and slowing down the aging process.
BUY NOW RAIN SOUL :
Rain International . rain soul Asia . rain soul North America . rain soul Canada . rain soul United States . rain soul USA . rain soul Europe . rain soul Germany . rain soul United Kingdom . rain soul Croatia . rain soul Hungary . rain soul Romania . rain soul Serbia . rain soul Slovakia . rain soul Asia . rain soul China . rain soul Hong Kong . rain soul Indonesia . rain soul Japan . rain soul Malaysia . rain soul Philippines . rain soul Singapore . rain soul Brunei . rain soul Taiwan . rain soul Thailand . rain soul Shanghai
BUY NOW RAIN CORE :
Rain International . rain core Asia . rain core North America . rain core Canada . rain core United States . rain core USA . rain core Europe . rain core Germany . rain core United Kingdom . rain core Croatia . rain core Hungary . rain core Romania . rain core Serbia . rain core Slovakia . rain core Asia . rain core China . rain core Hong Kong . rain core Indonesia . rain core Japan . rain core Malaysia . rain core Philippines . rain core Singapore . rain core Brunei . rain core Taiwan . rain core Thailand . rain core Shanghai
How to order rain soul . how to buy rain soul . buy rain soul . rain nutrition . rain soul how to buy . be a rain soul distributor .
The supplier abide the theory of "quality the basic, trust the first and management the advanced" so that they can ensure a reliable product quality and stable customers.