New Fashion Design for Lyophilized royal jelly powder Factory from Tunisia
New Fashion Design for Lyophilized royal jelly powder Factory from Tunisia Detail:
[Products Name] Royal jelly powder,Lyophilized royal jelly powder
[Specification] 10-HDA 4.0%, 5.0%, 6.0%, HPLC
[Gerneral feature]
1. Low antibiotics, Chloramphenicol< 0.1ppb
2.Organic certified by ECOCERT, according to EOS & NOP organic standard;
3.100% pure with no additives;
4. More easily absorbed into the body than fresh royal jelly
5. Can be easily produced into tablets.
[Our advantages]
- 600 bee farmers, 150 units of bee-feeding groups located in natural mountains;
- Organic certificated by ECOCERT;
- NON-antibiotics, widely exported to Europe;
- Health Certificate, Sanitary Certificate and Quality Certificate are available.
[Lyophilized technology]
Lyophilized technology, also known as Freeze-drying, it is a dehydration process typically used to maintain activity of all nutrition ingredients in royal jelly, also to make the royal jelly convenient for transport. Freeze-drying works by freezing the material and then reducing the surrounding pressure to allow the frozen water in the material to sublimate directly from the solid phase to the gas phase. This technology can maintain all activity of nutrition ingredient.
Lyophilized royal jelly powder is processed directly from fresh royal jelly.
3kgs fresh royal jelly is used to make 1kg lyophilized royal jelly powder.
During all the production process, there is no additives.
[Packing]
5kg/bag, 25kgs/drum
1kg/bag, 20kgs/carton
Main indices of physical and chemic in Lyophilized royal jelly
| Ingredients Indices | Lyophilized royal jelly | Standards | Results |
| Ash | 3.2 | <5 | Complies |
| Water | 4.1% | <7% | Complies |
| Glucose | 43.9% | <50% | Complies |
| Protein | 38.29% | >33% | Complies |
| 10-HDA | 6.19% | >4.2% | Complies |
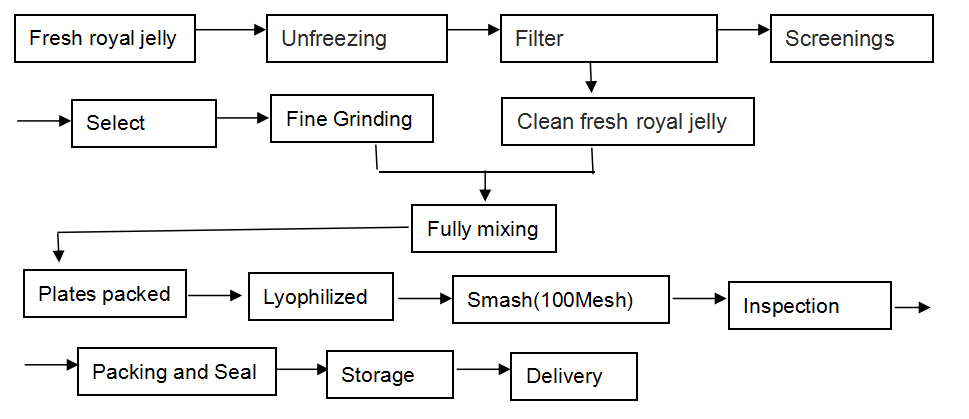
[Our work flow]
Our Lyophilized Royal Jelly Powder is produced in this way: we lyophilize the fresh royal jelly by advanced freeze-drying facilities without losing any nutritional ingredients, reserving the natural ingredients in utmost, and then make them into the form of powder, for any food additives are not needed to add.
The raw material we use is the natural fresh royal jelly which is up to the export standard . We process our products strictly according to export standard. Our workshop is up to the requirements of GMP.
Royal Jelly powder has been selected as drug excipients by many European and American pharmaceutical producing enterprises.Meanwhile it is applies to health food and cosmetics industries.
[Quality control]
Traceability record
GMP standard production
Advanced inspection equipment
[Function]
1.Enhances the immune system
2.Promotes wound healing
3.Has antitumor/anticancer properties
4.Lowers cholesterol levels
5.Increases fat metabolism
6.Is a powerful antioxidant
7.Regulates blood sugar levels
[Applications]
It’s widely used in health tonic, health pharmacy, hairdressing and cosmetic area, and mainly was applied in capsules, troche and oral liquids etc.
Product detail pictures:

Related Product Guide:
New Fashion Design for Lyophilized royal jelly powder Factory from Tunisia , The product will supply to all over the world, such as: , , ,
https://diabetessugarcontrol.mysocialtoolkit.com/
https://www.davegsmith.com/
https://davesmith.ludaxx.com
Powerful Antioxidant, How it Functions for
To conclude, usage of F21, PEARL and KONLI herbal tea for wellness physical fitness must be motivated because of their various synergistic results.
F21 is an All Natural Sugar Blocker that helps limit your blood sugar absorption. It consists of different compounds found to have various health benefits, such as L-Arabinose, Coriolus Versicolor Polysacchride, Konjac-Mannan, Magnesium Stearate, Mint flavor: Menthol and Natural Colors. F21 not only does it help promote weight loss, (PSK) boosts your immune system response. In fact, for every gram of F21, you can block up to 20 grams of sugar (sucrose). The formula not only helps promote weight loss, it benefits the digestive system by allowing the blocked sucrose to support beneficial probiotic bacteria while the polysaccharide (PSK) boosts your immune system response.
Contact
Dave Smith https://www.davegsmith.com/
Diabetes control :
00:00:05 Diabetes control
00:00:04 loseweight
00:00:03 Diabetes control
00:00:02 Herbs for diabetes
00:00:01 Diabetes control
Milk Thistle (Silybum marianum) is a European herb, with more than 2000 years of human consumption, used traditionally for liver disease, but is it actually effective? Find out here!
Scientific Literature:
Bosch-Barrera, J., & Menendez, J. A. (2015). Silibinin and STAT3: A natural way of targeting transcription factors for cancer therapy. Cancer Treatment Reviews, 41(6), 540-546. – https://www.ncbi.nlm.nih.gov/pubmed/25944486
Bosch-Barrera, J., Sais, E., Cañete, N., Marruecos, J., Cuyàs, E., Izquierdo, A., … Menendez, J. A. (2016). Response of brain metastasis from lung cancer patients to an oral nutraceutical product containing silibinin. Oncotarget, 7(22), 32006–32014. – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5077992/pdf/oncotarget-07-32006.pdf
Freedman, N. D., Curto, T. M., Morishima, C., Seeff, L. B., Goodman, Z. D., Wright, E. C., … the HALT-C Trial Group. (2011). Silymarin use and liver diease progression in the HALT-C trial. Alimentary Pharmacology & Therapeutics, 33(1), 127–137. – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3490214/pdf/nihms415758.pdf
Freitag, A. F., Cardia, G. F. E., da Rocha, B. A., Aguiar, R. P., Silva-Comar, F. M. de S., Spironello, R. A., … Cuman, R. K. N. (2015). Hepatoprotective effect of silymarin (Silybum marianum) on hepatotoxicity induced by acetaminophen in spontaneously hypertensive rats. Evidence-Based Complementary and Alternative Medicine : eCAM, 2015, 538317. – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4363982/pdf/ECAM2015-538317.pdf
Hong, M., Li, S., Tan, H. Y., Wang, N., Tsao, S.-W., & Feng, Y. (2015). Current status of herbal medicines in chronic liver disease therapy: The biological effects, molecular targets and future prospects. International Journal of Molecular Sciences, 16(12), 28705–28745. – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4691073/pdf/ijms-16-26126.pdf
Kawaguchi-Suzuki, M., Frye, R. F., Zhu, H.-J., Brinda, B. J., Chavin, K. D., Bernstein, H. J., & Markowitz, J. S. (2014). The effects of milk thistle (Silybum marianum) on human cytochrome P450 activity. Drug Metabolism and Disposition, 42(10), 1611–1616. – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4164972/pdf/dmd.114.057232.pdf
Kumar, R., Deep, G., & Agarwal, R. (2015). An overview of ultraviolet B radiation-induced skin cancer chemoprevention by silibinin. Current Pharmacology Reports, 1(3), 206–215. – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4471873/pdf/nihms-672250.pdf
Lazzeroni, M., Guerrieri-Gonzaga, A., Gandini, S., Johansson, H., Serrano, D., Cazzaniga, M., … & Bonanni, B. (2016). A presurgical study of oral silybin-phosphatidylcholine in patients with early breast cancer. Cancer Prevention Research, 9(1), 89-95. – https://cancerpreventionresearch.aacrjournals.org/content/canprevres/9/1/89.full.pdf
Schrieber, S. J., Hawke, R. L., Wen, Z., Smith, P. C., Reddy, K. R., Wahed, A. S., … Fried, M. W. (2011). Differences in the disposition of silymarin between patients with nonalcoholic fatty liver disease and chronic hepatitis C. Drug Metabolism and Disposition, 39(12), 2182–2190. – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3226378/pdf/zdd2182.pdf
Surai, P. F. (2015). Silymarin as a natural antioxidant: An overview of the current evidence and perspectives. Antioxidants, 4(1), 204–247. – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4665566/
Yang, Z., Zhuang, L., Lu, Y., Xu, Q., & Chen, X. (2014). Effects and tolerance of silymarin (milk thistle) in chronic hepatitis C virus infection patients: A meta-analysis of randomized controlled trials. BioMed Research International, 2014, 941085. – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4163440/pdf/BMRI2014-941085.pdf
Wu, J.-P., Tsai, C.-C., Yeh, Y.-L., Lin, Y.-M., Lin, C.-C., Day, C. H., … Huang, C.-Y. (2015). Silymarin accelerates liver regeneration after partial hepatectomy. Evidence-Based Complementary and Alternative Medicine : eCAM, 2015, 603529. – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4539063/pdf/ECAM2015-603529.pdf
Also, be sure to check us out at -
Subscribe – https://www.mikethecaveman.com
Like – https://www.facebook.com/paleoprimal.longisland
Follow – https://www.instagram.com/mikethecaveman
Like – https://www.twitter.com/mikethecaveman
Become a Patron – https://www.patreon.com/mikethecaveman
 By from -
By from -
 By from -
By from -