High Performance Lyophilized royal jelly powder Supply to Cape Town
High Performance Lyophilized royal jelly powder Supply to Cape Town Detail:
[Products Name] Royal jelly powder,Lyophilized royal jelly powder
[Specification] 10-HDA 4.0%, 5.0%, 6.0%, HPLC
[Gerneral feature]
1. Low antibiotics, Chloramphenicol< 0.1ppb
2.Organic certified by ECOCERT, according to EOS & NOP organic standard;
3.100% pure with no additives;
4. More easily absorbed into the body than fresh royal jelly
5. Can be easily produced into tablets.
[Our advantages]
- 600 bee farmers, 150 units of bee-feeding groups located in natural mountains;
- Organic certificated by ECOCERT;
- NON-antibiotics, widely exported to Europe;
- Health Certificate, Sanitary Certificate and Quality Certificate are available.
[Lyophilized technology]
Lyophilized technology, also known as Freeze-drying, it is a dehydration process typically used to maintain activity of all nutrition ingredients in royal jelly, also to make the royal jelly convenient for transport. Freeze-drying works by freezing the material and then reducing the surrounding pressure to allow the frozen water in the material to sublimate directly from the solid phase to the gas phase. This technology can maintain all activity of nutrition ingredient.
Lyophilized royal jelly powder is processed directly from fresh royal jelly.
3kgs fresh royal jelly is used to make 1kg lyophilized royal jelly powder.
During all the production process, there is no additives.
[Packing]
5kg/bag, 25kgs/drum
1kg/bag, 20kgs/carton
Main indices of physical and chemic in Lyophilized royal jelly
| Ingredients Indices | Lyophilized royal jelly | Standards | Results |
| Ash | 3.2 | <5 | Complies |
| Water | 4.1% | <7% | Complies |
| Glucose | 43.9% | <50% | Complies |
| Protein | 38.29% | >33% | Complies |
| 10-HDA | 6.19% | >4.2% | Complies |
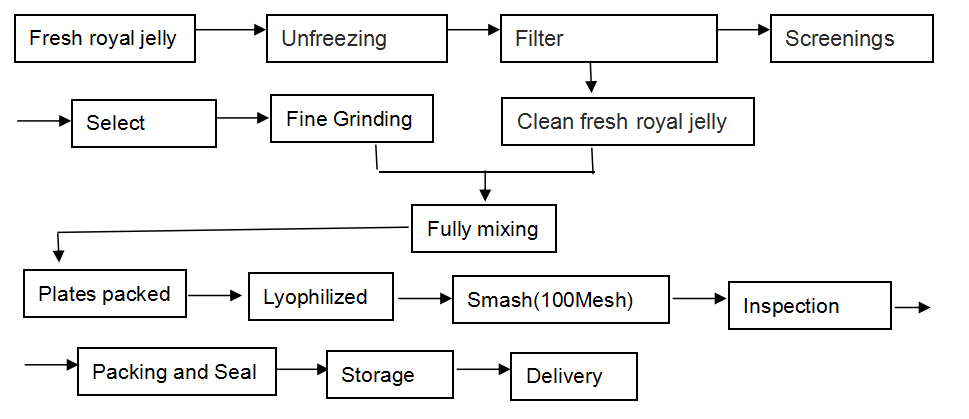
[Our work flow]
Our Lyophilized Royal Jelly Powder is produced in this way: we lyophilize the fresh royal jelly by advanced freeze-drying facilities without losing any nutritional ingredients, reserving the natural ingredients in utmost, and then make them into the form of powder, for any food additives are not needed to add.
The raw material we use is the natural fresh royal jelly which is up to the export standard . We process our products strictly according to export standard. Our workshop is up to the requirements of GMP.
Royal Jelly powder has been selected as drug excipients by many European and American pharmaceutical producing enterprises.Meanwhile it is applies to health food and cosmetics industries.
[Quality control]
Traceability record
GMP standard production
Advanced inspection equipment
[Function]
1.Enhances the immune system
2.Promotes wound healing
3.Has antitumor/anticancer properties
4.Lowers cholesterol levels
5.Increases fat metabolism
6.Is a powerful antioxidant
7.Regulates blood sugar levels
[Applications]
It’s widely used in health tonic, health pharmacy, hairdressing and cosmetic area, and mainly was applied in capsules, troche and oral liquids etc.
Product detail pictures:

Related Product Guide:
We stay with the basic principle of "quality initially, services first, steady improvement and innovation to fulfill the customers" for your management and "zero defect, zero complaints" as the quality objective. To perfect our company, we give the goods while using the good high-quality at the reasonable selling price for High Performance Lyophilized royal jelly powder Supply to Cape Town , The product will supply to all over the world, such as: Liverpool, Costa Rica, Slovenia, We have constructed strong and long co-operation relationship with an enormous quantity of companies within this business in Kenya and overseas. Immediate and specialist after-sale service supplied by our consultant group has happy our buyers. Detailed Info and parameters from the merchandise will probably be sent to you for any thorough acknowledge. Free samples may be delivered and company check out to our corporation. n Kenya for negotiation is constantly welcome. Hope to get inquiries type you and construct a long-term co-operation partnership.
Lemon Poppy Muffins Made with Stevia Extract
~*~*~*~*~*~ INGEDIENTS ~*~*~*~*~*~*~*~*~
JAJA Stevioside aka 95% Steviol Glycosides 100% Pure Stevia Extract – 1 Teaspoon
Lemon Juice – 1/2 cup
Sugarless Vanilla Extract – 1 fluid oz
Target Organic Flour – 3 Cups
Organic Eggs – 6
Poppy Seeds – 1/2 Cup
Baking Soda – 1.5 Teaspoons
Baking Powder – 3 teaspoons
* Using Target Brand “Simply Balanced” All Purpose Unbleached Flour – Get it at Target
* 1 oz Vanilla Extract made without sugar – Get it at any supermarket, various dollar stores, ethnic markets
* Real Lemon Lemon Juice – Get it at any supermarket
* Organic Eggs – Get it at any supermarket, Costco, BJ’s Target, WallMart, etc.
* Arm and Hammer Baking Soda – Get it at any supermarket, Costco, BJ’s Target, WallMart, etc.
* Rumford Brand Baking Powder NON GMO – Get it at any supermarket
* Organic Coconut oil – Get it at any supermarket, Costco, BJ’s, health food store, etc.
* JAJA Stevioside get it at https://emperorsherbologist.com/stevioside.php
www.whitestarcafe.com. White Star Cafe is a Modern Bistro and Artisanal Bakery. We are the country’s first restaurant to be Gluten, Soy, Corn, Treenut* (*except coconut), Peanut, Fish and Shellfish free, and also 100% Non-GMO and mostly Organic. We offer breakfast, lunch and dinner, as well as organic coffee, including Cuban coffee Artisanal Gelato and handmade Bread. Our products are made from scratch using latex free gloves, many are available dairy and egg free and also vegan. Although it may be hard to believe, we do not use any artifical preservatives, colorings or flavors.
This is a honest and trustworthy company, technology and equipment are very advanced and the prodduct is very adequate, there is no worry in the suppliment.